Chakradhar Maddela*
Neonatologist, Department of Neonatology, ESIC Medical College, Sanath Nagar, Hyderabad, India
*Corresponding Author: Chakradhar Maddela, Neonatologist, Department of Neonatology, ESIC Medical College, Sanath Nagar, Hyderabad, India.
Received: June 28, 2022; Published: July 27, 2022
Citation: Chakradhar Maddela. “BCG Adverse Effects in Infants - Case Series”. Acta Scientific Paediatrics 5.8 (2022): 26-30.
Bacillus Calmette-Guerin (BCG) is widely using vaccination for prevention of tuberculosis in children globally. BCG adverse effects are uncommon. Common adverse effects include BCG abscess, non-suppurative and suppurative lymphadenitis and rarely BCG disseminated disease. Common therapeutic strategies for BCG complications include wait and watch policy, needle aspiration, antibiotic therapy and surgery. Correct guidelines when and how to manage BCG complications are not available. We came across four cases of BCG adverse effects recently. All four cases were noted, followed up and presenting as case series with literature review on how to treat.
Keywords: BCG; Vaccination; Complications; Management.
ATT: Antituberculous Treatment; BCG: Bacillus CalmetteGuerin; HCF: Health Care Facility; HCP: Health Care Personnel; LSCS: Lower Segment Caesarean Section; OPD: Outpatient Department; USS: Ultrasound Scan
Bacillus Calmetti Gereine (BCG) is one of the most widely used vaccine for immunization in infants globally. It’s use intended to protect against tuberculosis infection and its dissemination. BCG vaccine protection against tuberculosis widely varies and argued by many. Its role expected in protecting miliary tuberculosis, neuro-tuberculosis [1] and shown extended protection in children from all-cause mortality and from leprosy [2]. Commonly used BCG strains for vaccination purpose are Danish 1331, Japanese and Russian strains. The abnormal adverse BCG response with large cold abscess formation depends on technique of vaccine administration and BCG strain used.
At our health care facility outpatient clinic, we came across four cases of large BCG abscesses in infants on different days and from different places recently. All four cases were noted with case details and followed up.
All four cases were infants, of which one infant was female child of 9 months old and other 3 cases were male infants.
Nine month old female baby brought for consultation for a swelling at BCG injection site. Baby delivered at full term by spontaneous vaginal delivery at a health care facility (HCF). It was a non consanguineous marriage of young couple. Antenatal, intranatal and postnatal history was uneventful. Baby vaccinated at birth by healthcare personnel (HCP). Baby had vaccination up-to-date. Baby developed large swelling at the site of BCG injection on left arm at 1 month. The swelling was progressed and attained 3 × 3 cm and not regressed or burst to heal up even after 9 months of age. An ipsilateral axillary lymphadenitis, single, non-suppurative, measuring 2.5 × 2.5cm2 was present. No adverse reactions occured to any other vaccinations given at later date as per immunization schedule to the child. There was no history of recurrent infections or hospitalization prior to consultation. No family history of immunodeficiency or allergy. Baby was on breast feeds and weaning introduced in 6th month of life. Baby weighed 8.2kg at 9 months and her birth weight was 2.9kg. Keeping in mind the parental apprehension and unresolved abscess and local non-suppurative lymphadenitis, infant given initially oral amoxicillin-clavulanate for a week without any response. Infant responded to oral INH and Rifampicin therapy with complete resolution.
Seventy-two days old male baby brought to our outpatient clinic for abnormal swelling at BCG injection site. Baby born to non-consanguineous young couple and delivered by lower segment caesarean section (LSCS) at a health care facility. Baby vaccinated at birth with BCG, oral polio vaccine and hepatitis B at HCF by HCP. Antenatal, intranatal and post-natal history was uneventful. There was no history of recurrent infections or hospitalization prior to consultation. Baby was on exclusive breast feeds. Swelling at BCG injection site noticed after 1month of injection, gradually increased in size and attained present size of 3. 4 × 2 cm, soft, nontender, fluctuant, well-defined borders and negative translucency test. Ultrasound scan revealed circumscribed lesion with anechoic central area and internal echogenic spots and showed no increased vascularity on colour Doppler. Baby was on follow up for wait and watch policy. The abscess was spontaneously drained after 3 days at home and later resulted in formation of large scar.
Sixty-four days old male baby brought to our OPD for large swelling at BCG injection site on left arm by attendants. It was a full term LSCS institutional delivery to a non-consanguineously married young mother. Pregnancy was uneventful. Baby was on top feeds for first 3 days followed by on breast feeds. Baby was vaccinated with BCG injection of left arm, OPV and Hepatitis B. Baby was well and up-to-date vaccinated. BCG abscess noticed on left arm measuring 2.7 × 1.8 cm. The abscess was drained with needle and syringe by HCP at her village 3 days prior to consultation and refilled partially. Swelling was showing portal of entry into abscess with needle, margins well marked, non-tender, fluctuant and translucency test negative. Mother revealed the aspirated content was pearly white liquid.

Figure 1: Abnormal BCG response in three infants. All three cases are showing large lesions with cold abscesses. Case 1 measured 3 × 3 cm Case 2 measured 3.4 × 2.3cm and Case 3 -partially drained by local HCW and measured 2.7 × 1.8 cm.
Four-month-old male child brought to our outpatient clinic with history of drained BCG abscess by health care personnel outside and showing scar of 2.9 × 2.6 cm on left arm towards acromion instead of towards deltoid insertion and ipsilateral, solitary, axillary lymphadenitis measuring 3.7 × 2.3 × 1.57cm and wall thickness of 1.05cm with increased peripheral vascularity on colour Doppler ultrasound scan. It is non-suppurative with no inflammatory signs clinically. BCG was given at birth by HCP. No other significant findings in history and on examination. Child’s total leukocyte count was 9,800 per cu.mm with neutrophils 49%, lymphocytes 44% and ‘C’ reactive protein 9.1mg/L. Child prescribed antibiotics (amoxicillin-clavulanate) in view of outside needle drainage and peripheral rim of increased vascularity of axillary lymph node on colour Doppler USS. Child is still on follow up.
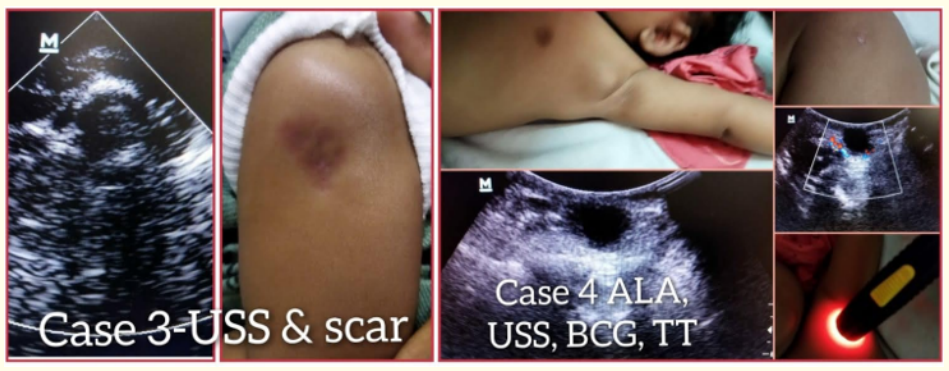
Figure 2: Showing case 3 ultrasound scan image and large BCG scar and case 4 showing solitary axillary lymphadenitis (left top), BCG scar (right top), ultrasound scan images and translucency test of lymphadenitis.
All neonatal infants receive BCG vaccine at birth in India under Universal Immunization Programme and globally in most of the countries as well. Normal BCG response includes formation of a wheal of > 5 mm immediately after the vaccination followed by papule formation by 2 weeks, further enlargement results in formation of nodule by 4 weeks. This is followed by pustule formation by 6 weeks and later self drains and results in healing with scar formation between 6-12weeks. Abnormal attainment in size with large abscess formation and local lymphadenitis can be caused by improper technique of administration of vaccine, type of vaccine strain used, age and immune status of the infant [3]. BCG lymphadenitis develops by 8weeks after injection administration [4]. Adverse reactions are more often seen with Danish strain (more effective) and least common with Russian strain. Danish strain previously widely used in India and is now replaced by Russian strain by the vaccine makers Serum Institute of India. All the four cases were vaccinated with BCG Russian strain and manufactured by the Serum institute of India. Common BCG injection complications include abscess formation at injection site (1/3 of complications), local/axillary lymphadenitis (2/3 of complications) with or without suppuration, and BCG disease (disseminated) including osteomyelitis. These complications present usually within 6months of injection (Venkatraman A 2015).
Our case 1 and 4 had BCG abscesses and axillary lymphadenitis whereas case 2 and 3 were presented with BCG abscesses. Case 2 spontaneously ruptured and healed with large scar. Case 3 and case 4 were drained outside by HCP and were prescribed antibiotics. Case 3 partially drained outside. Only case 1 was treated with ATT and resolved completely. Case 4 had BCG scar but having nontender, non-suppurative axillary lymphadenitis and on follow up.
BCG vaccination - In newborns, BCG should be given by the trained vaccinator with correct dose of 0.05ml (0.1ml in children) by intradermal route with 26G needle at insertion of deltoid on left upper arm.
Natural course of BCG response and its variants: Normal BCG response after injection was already discussed above in discussion section. BCG injection has got both therapeutic and diagnostic value. BCG reaction is said to be accelerated if an induration of > 5mm at the injection site occurs within 24 - 72 hours, hyper-accelerated if the same induration noticed between 6-12 hours and delayed response if same induration response occurs between 72hours to 2weeks [5]. These reactions may indicate latent tuberculous infection or active tuberculous infection and warrants further evaluation. BCG adverse reactions includes 1. local BCG abscess, 2. local non-suppurative lymphadenitis, 3. local suppurative lymphadenitis and 4. disseminated BCG disease [6].
BCG adverse reactions common in the following circumstances - 1. When BCG wrongly administered by subcutaneously 2. When given on upper 1/3 of arm 3. When BCG given to newborn on day one or within 1st week of life 4. When BCG vaccine vial contains less live bacteria and more killed BCG bacteria 5. When the antigen dose increased 6. Preterm infants < 37weeks gestational age experience 6times more risk of developing local lymphadenitis 7. Three times increased risk of lymphadenitis was noticed when BCG administered on right arm instead of on left arm [7].
There are no conflicts of interest for this study.
There are no sponsors for this study.
0000-0001-7475-0777.
Copyright: © 2022 Chakradhar Maddela. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ff
© 2024 Acta Scientific, All rights reserved.