Adwina Nurlita Kusuma Wardhani and Retno Asih Setyoningrum*
Department of Child Health, Faculty of Medicine Airlangga University, Surabaya, Indonesia.
*Corresponding Author: : Retno Asih Setyoningrum, Department of Child Health, Faculty of Medicine Airlangga University, Surabaya, Indonesia.
Received: January 19, 2022; Published: January 31, 2022
Citation: Adwina Nurlita Kusuma Wardhani and Retno Asih Setyoningrum. “Profile of Xpert MTB/RIF in Children with Suspected Tuberculosis in Tertiary Hospital in Surabaya, Indonesia”. Acta Scientific Paediatrics 5.2 (2022): 21-26.
Background: The accurate diagnosis of childhood tuberculosis remains a major challenge. Molecular diagnostic tests with Xpert MTB/RIF are more rapid and cost-effective compared to the conventional culture techniques used for diagnosis and drug-resistance testing. This study aims to describe the characteristics of children with suspected tuberculosis tested with Xpert MTB/RIF in a tertiary hospital in Surabaya, Indonesia.
Methods: A descriptive study was conducted on children with suspected tuberculosis in an academic teaching hospital in Surabaya, Indonesia between January 2016 and December 2020. Clinical materials were submitted to the laboratory for examination-including sputum, pleural fluid, and gastric aspirate-and the Xpert MTB/RIF test was performed. Outcomes described as Mycobacterium Tuberculosis (MTB) detected, MTB not detected, rifampicin-sensitive, and rifampicin-resistant.
Results: Among 887 subjects during five years study period, 253 (28,5%) were sputum samples, with others from gastric aspirate, cerebrospinal fluids, feces, material from abscesses, and other biopsy specimens or resected tissue. The total number of MTB detected was 155 (17,5%) with the majority collected from 109 (70,3%) sputum samples, and the highest number of MTB Detected was found in age 10-18 years old 105/155 (67,7%). Drug sensitivity test showed 13 (8,3%) rifampicin resistance with all samples were sputum, while 142 (91,6%) subjects showed no rifampicin resistance.
Conclusions: Sputum was the largest sample collection of children tested with Xpert MTB/RIF, it showed a considerable amount of MTB positivity, with the majority aged 10-18 years old. Drug sensitivity test showed rifampicin-resistant results came from sputum samples.
Keywords: Tuberculosis; Children; Xpert MTB/RIF; Profile.
Tuberculosis (TB) is one of the major causes of pediatric mortality, with half a million cases every year in the world [1,2]. The number of Indonesian pediatric TB cases fluctuates each year, [3] indicating the need to evaluate the pediatric TB diagnosis’ quality in Indonesia.
World Health Organization recommends Xpert MTB/RIF which could detect the Mycobacterium tuberculosis (MTB) and rifampicin-resistance under 100 minutes [2]. A study [5] found that it could detect 90.3% MTB. Another study [6] found that children’s sputum provides high accuracy. This study aims to describe the profile of suspected pediatric TB tested with Xpert MTB/RIF in a tertiary hospital in Surabaya, Indonesia.
A retrospective descriptive study was performed in an academic teaching hospital in Surabaya, Indonesia. The accessible population was all children with suspected TB in the same hospital who had undergone a rapid molecular test of Xpert MTB/RIF in the period of study. Children aged 0 - 18 years who had incomplete medical record data were excluded from this study. A total of 887 children were recruited in this study. Ethical approval for this study was obtained from The Ethics Committee of the Faculty of Medicine, Universitas Airlangga – Dr. Soetomo Academic General Hospital with number 1037/KEPK/III/2019.
This study uses secondary data from the results of rapid molecular test Xpert MTB/RIF in the period of January 2016 – December 2020. Data were collected from the medical records in the Multidrug Resistance Tuberculosis outpatient clinic of the hospital. The data were based on a variety of specimens submitted to the laboratory for examination, including sputum, pleural fluid, and gastric aspirate from the hospital and outside the hospital. Xpert MTB/RIF was used as the diagnostic test with the outcomes described as MTB detected, MTB not-detected, rifampicin-sensitive, and rifampicin-resistant.
Total sampling was conducted for this study. After population recruitment, it is then classified into 4 groups of variables: demographic data, sample characteristics, Xpert MTB/RIF results, and rifampicin resistance.
The data were analyzed using a descriptive statistical test with version 22.0: SPSS Software, IBM, Chicago, IL, USA. Statistical analysis was based on the calculation of frequency distribution.
A total of 887 data were obtained with the characteristic based on the clinical sample presented in Table 1.
Table 1 shows that, from 2016 until 2020, the largest samples collected for TB diagnosis are sputum (28.5%) followed by mucus (24.4%) and saliva (16.6%). Nevertheless, samples from stools and biopsy tissues were increasing in the last two years.
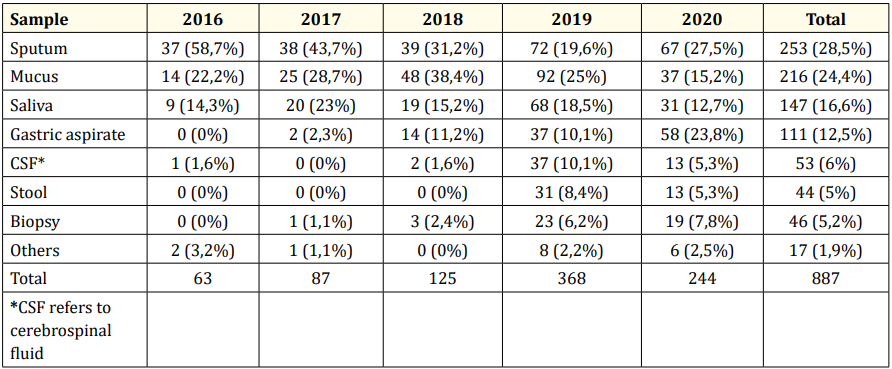
Table 1: Clinical sample characteristics.
Among 887 participants, MTB Detected and MTB Not Detected were 155 patients and 732 patients respectively. More than half of participants who had MTB Detected are found in the 10 - 18 years age group, classified into rifampicin-sensitive (66.2%) and rifampicin-resistant (84.6%). Despite that, the highest number of MTB Not Detected is found in this age group as well.
Table 3 describes the results of the Xpert MTB/RIF test based on age and clinical specimens. It is found that MTB Detected is predominant in the 10 - 18 years group (n = 101). Primarily, the clinical samples were from sputum (n = 84).
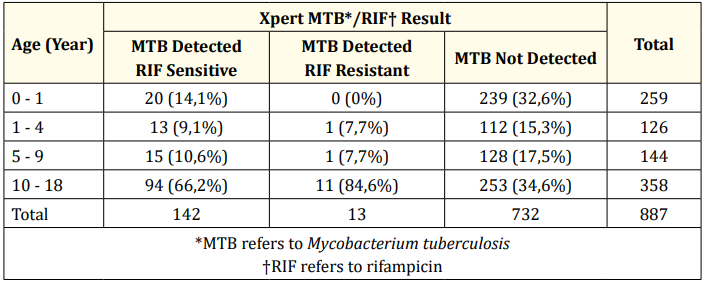
Table 2: Xpert MTB/RIF result based on age.
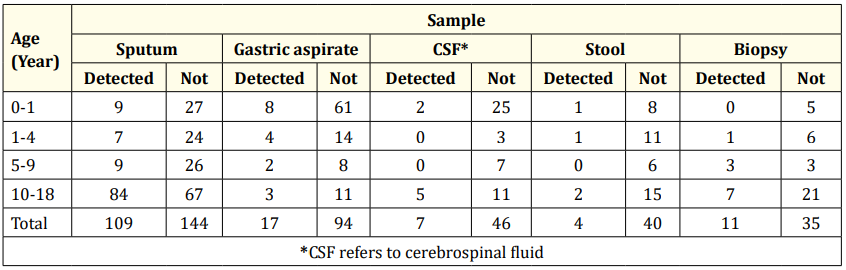
Table 3: Xpert MTB/RIF results based on age and clinical specimen.
As seen from Figure 1, from 155 participants with MTB Detected results, almost all of the subjects [142 (91.6%)] have rifampicin-sensitive and only a few subjects [13 (8.4%)] have a rifampicin-resistant type. All samples from 13 patients, who had rifampicin-resistant MTB, were from sputum. Thus, concurrent with rifampicin-sensitive group examination, the majority of the results were from sputum [96 (67.6%)] followed by gastric aspirate [17 (11.97%)] and biopsy tissue [11 (7.7%)]. Figure 2 represents the results of sample collection in the rifampicin-sensitive group.
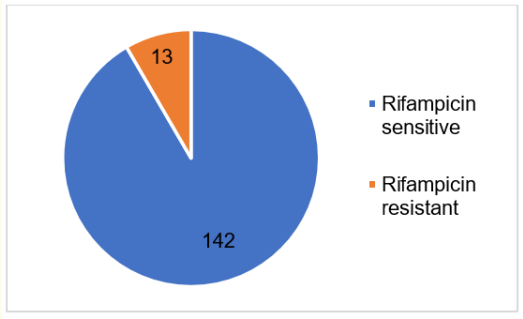
Figure 1: Rifampicin sensitivity result. Blue represents rifampicin sensitive. Orange represents rifampicin resistant.
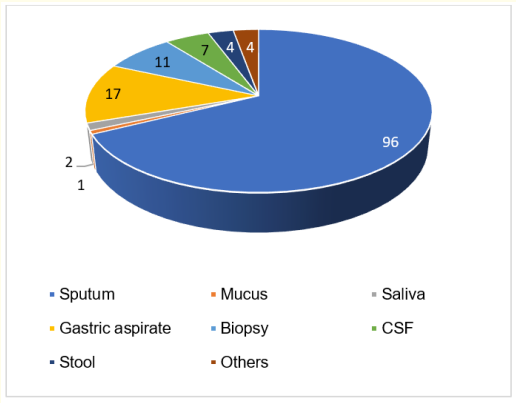
Figure 2: MTB rifampicin-sensitive result based on specimen. Most rifampicin-sensitive results came from sputum (n = 96), Followed by Gastric Aspirate (n = 17), Biopsy (n = 11), Cerebrospinal Fluid (CSF; n = 7), Stool (n = 4), others (n = 4), Saliva (n = 2), and Mucus (n = 1).
This study occurred by dint of early diagnosis obstacle especially in suspected pediatric TB patients which is crucial for TB management. Some conventional TB diagnosis instruments already existed, such as clinical approach with TB score, acid-fast bacillus examination, and MTB culture [7]. All of these had a weakness, including MTB culture, the gold standard for TB diagnosis. It takes six to eight weeks of the diagnostic process which might delay patients’ initial treatment. Sputum examination with Xpert MTB/RIF, a rapid molecular test using polymerase chain reaction DNA, provides faster and more sensitive results than the other conventional instruments [6].
Our findings showed that among 887 children with suspected TB who had been tested with Xpert MTB/RIF, there were 253 (28.5%) specimens from sputum. Concurrent with a study from Walusimbi., et al. [8] most samples come from the sputum of TB patients. The study also showed that Xpert MTB/RIF has high sensitivity (98 - 100%) on positive BTA and culture samples, although sensitivity on negative BTA samples is found to be only 43 - 70% [8]. Nicol., et al. [4] conducted a similar study in children and found that Xpert MTB/RIF sensitivity increased 27.8% with 98.8% specificity.
TB mainly occurs in the lung, yet extrapulmonary TB is also found in numerous cases. Bunyasi., et al. [9] revealed that samples from sputum have higher sensitivity and specificity than samples from gastric aspirate in diagnosis TB with Xpert MTB/RIF. The sensitivity was 26.7% and 22.6%, while specificity was 100% and 99.6% from sputum induction and gastric aspirate samples respectively [9]. Yet, a metanalysis done by Padmapriyadarsini., et al. [10] from 12 research, revealed that the sensitivity of Xpert MTB/RIF on gastric aspirate samples from TB pulmonary patients was 83.8% with 98.1% specificity. Simultaneously with a study by Hillemann., et al. [11] Xpert MTB/RIF examination on extrapulmonary samples (urine, biopsy tissue, pleural aspirate, cerebrospinal fluid, and stools) from TB patients have 77.3% sensitivity and 98.2% specificity in general. Exclusively, biopsy tissue has 69.0% sensitivity, and urine and stools have 100% sensitivity [11].
Walters., et al. [12] conducted a study in South Africa using Xpert MTB/RIF in extrapulmonary TB patients with the sample from stools. They found that sensitivity and specificity were unexpectedly high with 31,9% (95%CI 21,84 - 44,50) for sensitivity and 99,7% (95%CI 98,2 - 100) for specificity [12]. Another study from Lu., et al. [13] in China revealed that extrapulmonary TB examination from bronchoalveolar lavage has 72.9% and 98.7% for sensitivity and specificity respectively. However, Xpert MTB/RIF examination on several specimens for one patient reportedly does not increase the case detection rate [4]. Hence, in daily clinical practice, Xpert MTB/RIF test using two or more types of samples have to be considered regarding its risk and benefit since there are other diagnostic examination methods.
The results from our study showed that among 155 Xpert MTB/ RIF detected patients, 109 (70.3%) comes from sputum specimen, predominantly in the 10 - 18 years age group (n = 101). Similar to a study from Marais., et al. [14] teenagers tend to have a higher risk to be infected by TB, especially post-menarche female patients. This is due to children under ten years old generally having paucibacillary TB type, leading to a low MTB detected. Primary infection during teenage years is related to adult-type TB disease development, resulting in more sputum accumulation and usually gives positive examination results. This type could be developed by primary infection, endogen reactivation, and exogen reactivation. Therefore, the possibility of positive results from Xpert MTB/RIF is increased [15]. Marais., et al. [14] revealed that this type occurs in children above ten years post-primary infection.
The drug sensitivity test showed 13 (8,3%) rifampicin resistance with all samples were sputum, while 142 (91,6%) subjects showed no rifampicin resistance. Concurrent with our findings, Arega., et al. [16] revealed that in pediatric TB patients, 7.9% rifampicin-resistant MTB were positive. Yet another study, which was conducted in Indonesia, showed that among children with suspected pulmonary TB who tested positive Xpert MTB/RIF, 100% were rifampicin sensitive [17]. Positive results of MTB rifampicin resistance indicate that MTB bacteria possibly have a high resistance to rifampicin.
Since this study was retrospective research merely using secondary data, limitation in our study includes not all samples tested by culture MTB and incomplete medical record. Therefore, comprehensive analysis of TB contact history, total TST, anthropometry status, thorax x-ray result, anatomical location, medical history of TB, also HIV infection status were difficult to be done.
The largest sample collections of children who tested Xpert MTB/RIF were sputum, with a considerable amount of MTB positivity and majority from 10 - 18 years old age group. Drug sensitivity test showed all rifampicin-resistant results came from sputum samples.
Based on World Health Organization estimation in 2020, a 25% decrease in TB case detection over 3 months will result in a 13% increase in the number of deaths due to TB: delaying the progress of successful TB programs in the last 5 years. It is estimated that there will be an additional 1.4 million deaths in TB patients due to the consequences of COVID-19 in 2020 - 2025 [18].
Based on these results, further research was recommended to determine and compare diagnostic value between the rapid molecular test using Xpert MTB/RIF, the microscopic examination of Acid-Fast Bacilli, and the MTB culture using a cohort method; also identify more complete data related to positive MTB/RIF result. This is to succeed in the national target of reducing TB incidence and mortality by increasing the number of children diagnosed and treated, as well as investigation of household contacts to conduct TB prevention therapy at the right time.
Nill.
Nill.
Copyright: © 2022 Adwina Nurlita Kusuma Wardhani and Retno Asih Setyoningrum. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
ff
© 2024 Acta Scientific, All rights reserved.